Application Note:
Comparison of Transfection Reagents for pDNA and mRNA Delivery
BACKGROUND
Peripheral blood derived mononuclear cells (PBMCs) are important mediators involved in immune surveillance and are actively explored as the foundation of a wide range of therapies [1]. It is convenient to isolate the cells from patients and re-administer them after desired manipulations. There are intensive efforts to genetically modify the cells using vectors that permanently integrate their cargo into cellular genome. Transient expression of transgenes, on the other hand, is more desirable to exert some control over the expression kinetics[2]. Transient expression can be implemented with plasmid DNA (pDNA) or messenger RNA (mRNA) based expression systems [3]. Non-viral delivery of expression systems minimizes any effects on the host genome and leads to a safer intervention. Transfection reagents developed by RJH Biosciences are particularly suitable for this application since they are highly compatible with human cells and achieve elevated levels of delivery. Lipofection has been alternatively explored for modification of primary cells and a number of leading commercial reagents are based on lipid based reagents. This application note summarizes experience with transfection of PBMC (and suspension growing Jurkat Cells) with pDNA and mRNA, comparing commercial lipofection and polymer reagents to a leading reagent developed by RJH Biosciences.
MATERIALS and METHODS (general procedures)
RESULTS and DISCUSSION
To assess efficiency of the transfection reagents in PBMC, we used pDNA and mRNA based expression systems for Green Fluorescent Protein (GFP). Both expression systems were obtained from commercial vendors and GFP expression was investigated after 2 days. Optimal ratios recommended by vendors were used to form the complexes, as well as an excess ratio beyond the recommended range. As shown in Figure 1, GFP expression was readily obtained with ALL-Fect using the pDNA based expression system, which was superior to the lipofection reagents used here.
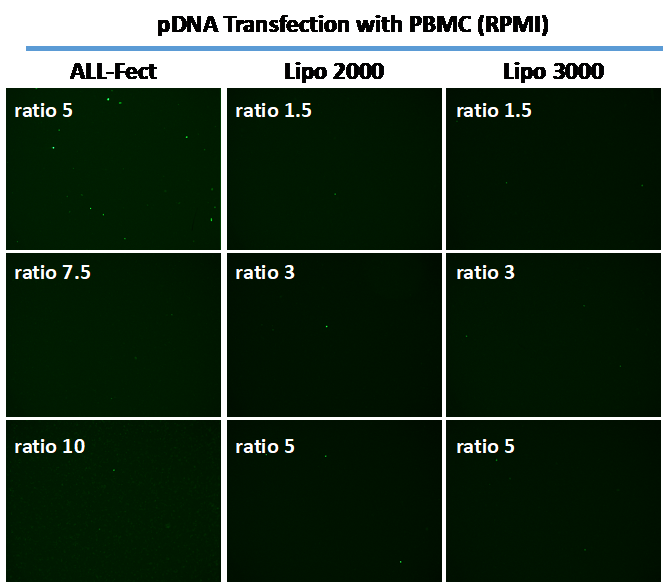
Figure 1. Typical transfections seen with PBMC and transfection reagents ALL-Fect (left), LipofectamineTM 2000 (middle) and LipofectamineTM 3000 (right). The cells were transfected with a GFP expressing pDNA using the indicated transfection reagents and GFP expression was visualized after 2 days with a fluorescence microscope. Optimal transfection with ALL-Fect was at ratio of 5 (carrier:pDNA).
In a follow-up experiment, PBMC were modified with mRNA using the same transfection reagents at the same ratios. The results are summarized in Figure 2. Similar to the results seen with pDNA in Figure 1, cells gave the best GFP expression with ALL-Fect reagent. There were no obvious signs of toxicity with the transfection reagents under the experimental conditions.
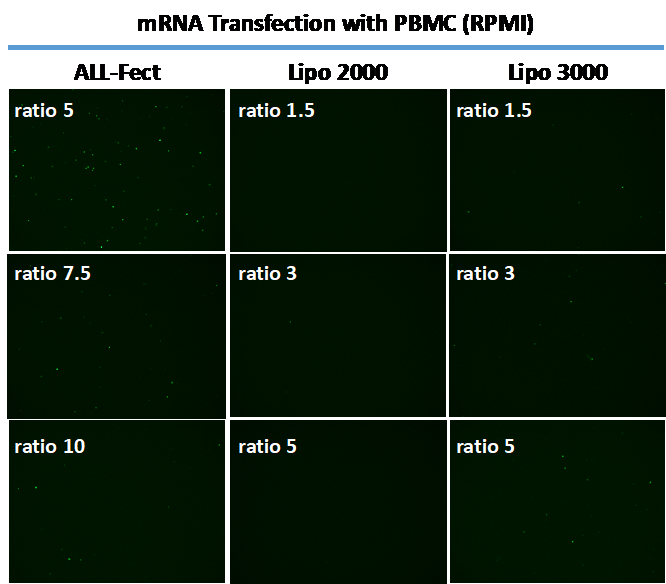
Figure 2. Typical transfections seen with PBMC and transfection reagents ALL-Fect (left), LipofectamineTM 2000 (middle) and LipofectamineTM 3000 (right). The cells were transfected with a GFP expressing mRNA using the indicated transfection reagents and GFP expression was visualized after 2 days with a fluorescence microscope. Optimal transfection with ALL-Fect was at ratio of 5 (carrier:pDNA).
A similar study was conducted with Jurkat T-cells. Due to more robust proliferation activity in these cells, they are more amenable for transfection, and they did not require activation. As can be seen in Figure 3 (next page), Jurkat cells were readily transfected with the chosen reagents and by using pGFP or mGFP. For cells transfected with the pGFP, jetOPTIMUSTM at carrier:pDNA ratios of 1 and 1.5 provided the highest GFP fluorescence per cell, while the percentage GFP-positive cells were equivalent between the two transfection reagents. Higher ratio of jetOPTIMUSTM (5) had reduced the cell numbers, indicating some toxicity. For cells transfected with the mGFP, ALL-Fect reagent provided the best transfection (based on both mean GFP fluorescence/cell and %GFP-positive population), while LipofectamineTM 3000 provided some transfection, unlike the jetOPTIMUSTM. Others have also employed PEI and Lipofectamine reagents for modification of Jurkat cells with transfection efficiencies of less than 5% in independent studies [4].
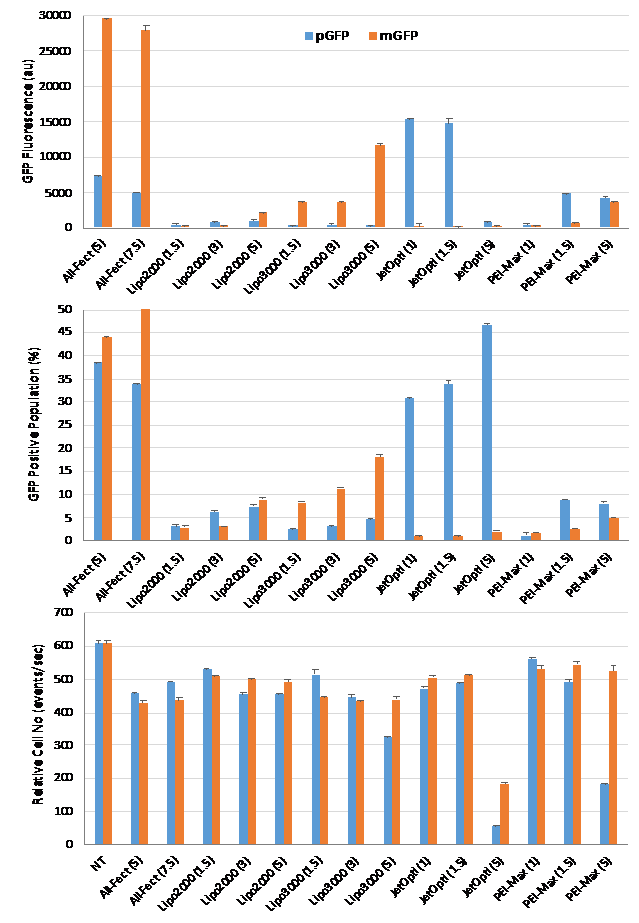
Figure 3. Typical transfection efficiencies seen with Jurkat T-cells and transfection reagents ALL-Fect, LipofectamineTM 2000 and LipofectamineTM 3000, JetOptimusTM and PEI MAXTM The cells were transfected with a GFP expressing pDNA (pGFP) or mRNA (mGFP) using the indicated transfection reagents. The specific ratios used to complex the pGFP or mGFP with the reagents are indicated in brackets. The complexes were prepared in RPMI1640 medium, given its routine use in cell culture. The GFP expression was quantitated by flow cytometry after 2 days. The results are summarized as mean GFP Fluorescence per cell (above), percentage of GFP-positive cell population (middle) and relative cell counts as a measure of cytotoxicity (bottom). Among the reagents tested, ALL-Fect optimally performed with both pDNA or mRNA based expression systems.
Benefits of RJH Transfection Reagents
REFERENCES

RJH manufactures a variety of transfection agents for your R&D needs. Download our catalogue for a list of our products.